Spontaneous neuronal regrowth and repair are not observed in adult spinal cords, making paralysis from spinal cord injury devastating and often permanent.
In a new study, Professor Samuel Stupp’s research team at Northwestern University created human spinal cord organoids (miniature organs derived from human induced pluripotent stem cells) to model different types of spinal cord injuries and test a promising new regenerative therapy.
While many researchers have developed human organoids to study the physiology and pathology of the spinal cord, Stupp’s group is using them to explore a more ambitious possibility: repairing paralyzing injuries in humans.
In an article published recently in Nature Biomedical Engineering, lead author Dr. Nozomu Takata and his colleagues demonstrated for the first time that human spinal cord organoids can reproduce key features of spinal cord injury in humans, including cell death, inflammation, and glial scarring (the creation of a dense mass of glial cells that acts as a physical and chemical barrier to nerve regeneration). Their treatment suppressed glial scar formation, reduced inflammation associated with injury and promoted significant axonal regeneration.
I interviewed Dr. Takata on the therapeutic which the lab calls “dancing molecules.” Dr. Takata told me he began his work in Japan, where he was among the first researchers to use principles of developmental biology to re-create organ formation in a Petri dish. After continuing his research in Chicago, at the Center for Regenerative Nanomedicine, he realized that the next logical step, after years of studying human organoids, was to create organoid models of traumatic injuries by damaging them and using them to validate a regenerative therapy.
Dr. Takata modelled injury in two ways: a simple laceration to test the resulting damage; and a contusion. The contusion model closely mimics the injuries people often sustain in car accidents or severe falls. Because the timing and force of the compression can be precisely controlled, the injury is highly reproducible in the lab. Both injury types produced hallmark effects of spinal cord trauma – cell death and the formation of a glial scar.
After establishing a mature spinal cord organoid, Dr. Takata wanted to examine the effect of subsequent spinal cord treatment using the lab’s platform of supramolecular therapeutic peptides called “dancing molecules.”
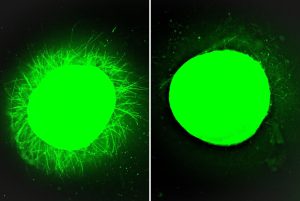
Fluorescent micrographs. Left: human spinal cord organoid treated with fast-moving “dancing molecules,” showing increased neurite outgrowth. Right: human spinal cord organoid treated with slow-moving “dancing molecules.” Neurite outgrowth is much more subdued than the organoid on the left. Images: Stupp Lab
What are dancing molecules?
First introduced in a 2021 Science paper by the Stupp lab, a single injection of dancing molecules therapy had been shown to harness molecular motion to trigger axonal growth, prevent glial scarring, and promote functional recovery in mice after traumatic spinal cord injuries (meaning they had recovered some of their locomotion after injury).
The term “dancing molecules” refers to the dynamic motion of molecules within a nanostructure. Although the molecules assemble into stable fibres, the hydrogen bonds and other non-covalent interactions that hold them together are relatively weak and transient. This allows the molecules to move while maintaining their overall nanostructure, enabling them to interact more effectively with receptors on the surface of cells.
In the interview, I also spoke to Dr. Liam Palmer, Research Professor of Chemistry and Director of Research at the Center for Regenerative Nanomedicine. “The molecules may pop up a little or shift slightly,” explained Dr. Palmer, “allowing them to reorganize, or ‘dance,’ to optimize their geometry for binding to cell surface receptors.” Dr. Palmer is a co-author of the paper.
The researchers also examined how the speed of this molecular motion affects signalling. By adjusting the strength of hydrogen bonding, they could fine-tune how freely the molecules moved. When the bonds were weaker, the molecules moved more rapidly, increasing opportunities to interact with receptors and improving cell signalling.
“By dialling back the hydrogen bonding, it allows the molecules to move around more, giving more opportunities for interaction with receptors. The cell signalling is better when the molecular motion is designed to be fast rather than slow,” said Dr. Palmer.
Once these supramolecular peptides engage cell receptors, they trigger regenerative processes such as reduced scar formation, reconnection of axons, repair of neural networks, and improvement of locomotion in vivo in preclinical animal models of spinal cord injury. The tissue regeneration after spinal cord damage was also seen in their human spinal cord organoid.
Producing a regenerative environment
One of the critical receptors that the dancing molecules bind to is called beta-1 integrin – it is one of many ways neurons interact with their environment. Once the molecules bind to this receptor, a cascade of signals that promotes regeneration begins.
Developmental biology inspired this insight into what would allow for a regenerative environment for the spinal cord. Prenatally, when growth of the spinal cord is rampant, there is higher expression of beta-1 integrin present on the neurons, whereas in adulthood, integrin expression declines significantly. This decline provides a clue as to why the adult spinal cord has limited to no ability to repair itself after injury.
“But if we use very bioactive integrin stimulation using our nanomaterials reported in the Science paper [in] 2021,” Dr. Takata said, “we may be able to help adult neurons grow again.”
Dr. Palmer describes the strategy more simply: “We’re essentially tricking the nervous system into thinking it’s in a regenerative environment.”
The first spinal cord injury model with an immune component
This research is also the first to incorporate microglia – a type of immune cell – into a central nervous system organoid injury model, simulating the inflammatory response seen in human traumatic spinal cord injury.
Dr. Takata explained that the contusion model of spinal cord injury involves mechanically compressing the human organoid tissue to create traumatic damage. “That causes cells to die, just like what would happen in a person who suffered a traumatic spinal cord injury. The dying cells is [sic] a message for the immune cells to be activated,” Dr. Takata said. Once activated, the microglia release factors that trigger astrocytes to form a glial scar, which is the dense tissue around the injury site. The astrocytes also secrete chondroitin sulfate proteoglycan, a chemical that inhibits neural regeneration.
By adding microglia, the organoid becomes a more biologically realistic platform for testing translational therapies, particularly because immune responses are one of the major pathological drivers of scar formation as barriers to regeneration after human spinal cord injury.
Clinical trials
For a person with a spinal cord injury, the researchers hope that a single injection of their dancing molecule therapy will allow for regeneration.
Within 24 hours of a severe spinal cord injury, neurosurgeons commonly perform decompression surgery to remove bone fragments and relieve pressure on the spinal cord. This procedure could also provide an ideal opportunity to administer the therapy directly to the injury site, potentially preventing the formation of the initial glial scar.
Human organoids provide one of the closest experimental models to the human spinal cord, making them an important step toward clinical translation. The researchers are currently working with the U.S. Food and Drug Administration to advance the therapy toward clinical trials. The agency has already granted the treatment Orphan Drug Designation.
“We think it’s still at least a year away,” Dr. Palmer said, “but we’re getting closer to a human trial. There are still many safety and efficacy studies underway, and we’re working through them now.”
Krystal Jacques
Latest posts by Krystal Jacques (see all)
- New paralysis treatment tested on injured human organoids is close to clinical trials - April 28, 2026
- A new one-and-done lifetime gene therapy treatment for fatal Hunter syndrome - January 19, 2026
- Brain-computer interface could offer solution to people with motor neuron degeneration - November 5, 2025








Comments