>
There are few things in Canada that grab national attention than a story related to hockey. News that surfaced last month that hockey legend Gordie Howe had received an unproven stem cell therapy for stroke continue to grab headlines in this country. In the past week alone, our office has fielded several media calls and requests from stroke patients for comment or recommendations on the controversial therapy. A second, unrelated story about Regenetek, a Winnipeg-based company selling an unproven MS therapy in India has once again put stem cell tourism back into the public sphere.
What I have found immensely challenging in the media coverage on the two stories is the lack of clarity offered. Oddly, in this respect, I seem to be in alignment with Regenetek, who wrote in a statement published yesterday by CBC (scroll to bottom of story for company statement) that “…the standards and protocols discussed in the media are poorly understood…”
But while Regenetek is focused on how their company and the protocol they purportedly developed and are tracking (as administered by “third party medical practitioners”) has been “mischaracterized”, I’m more concerned about what they are and are not saying to clarify the alleged “misinformation or misunderstanding”. Paul Knoepfler does a great job of highlighting the confusing statements made in the Gordie Howe case and by representatives of Regenetek. The statement published yesterday by CBC only further obfuscates the matter, as does the fact that the Regenetek website is now curiously under construction, offering no information at all. So what is it they’re hiding?
Here’s an example: If, as published in the statement to CBC indicates (bolding done by me to highlight areas of contradiction),
“Regenetek Research and its researchers do not provide medical treatment in connection with the clinical study. Further, the study was never intended to replace other medical treatment and individuals were directed to stay in contact with their treating physicians. We are scientists simply tracking the progress of participants who have chosen to attempt innovative therapy options and to have their results noted by us for scientific study.”
then the company is tracking patients who would seem to have chose a therapy “provided by third party medical practitioners”. Yet, their currently active (at time of publication) Regenetek Google+ page states that (again, bolding is my own),
“Regenetek is a private not-for-profit medical research company that is advancing research into regenerative medicine through therapeutic interventions, many of which the company has developed. The company has translated the science from clinical trials to offer leading edge therapies for the effective treatment of a wide range of chronic neurological diseases.”
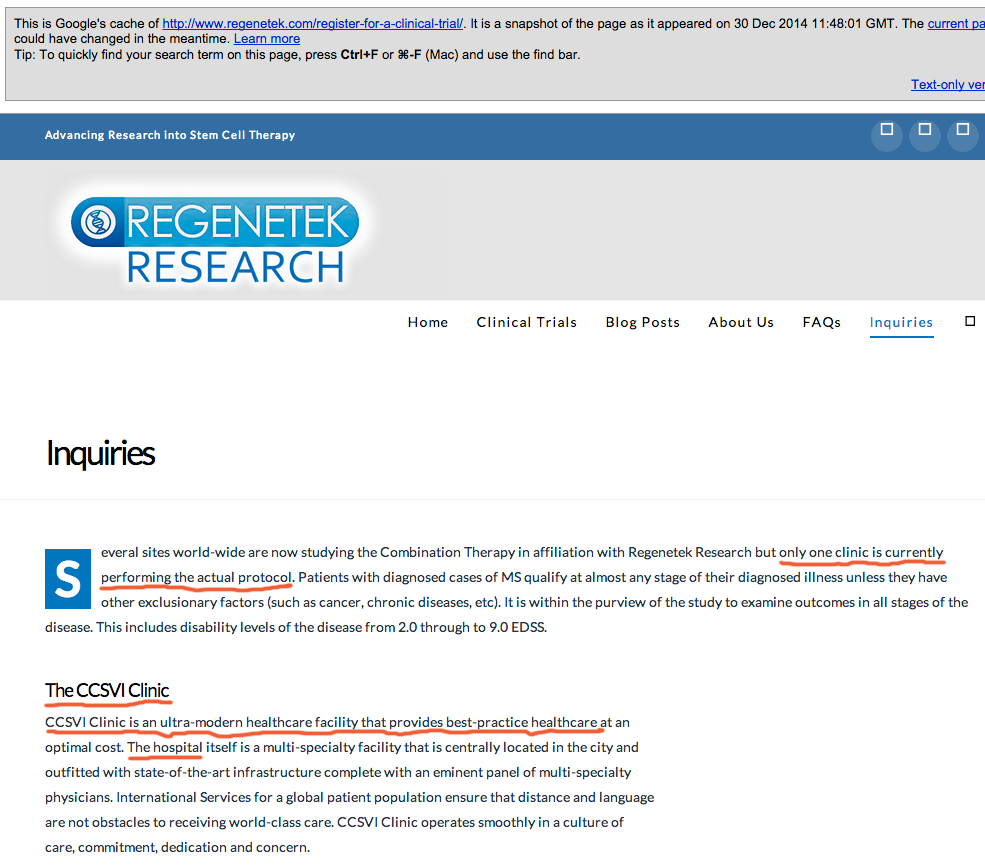
How is it that a company that is “simply tracking the progress of participants who have chosen an innovative therapy” is also involved in the development of the therapy and presented itself as offering “leading edge” “effective” treatment? It’s unclear what the relation is. As a second example, when I viewed the cached copy of the Regenetek website (Google cache from December 30, 2014; see image at bottom), it stated that
“CCSVI Clinic is an ultra-modern healthcare facility that provides best-practice healthcare at an optimal cost. The hospital itself is a multi-specialty facility that is centrally located in the city and outfitted with state-of-the-art infrastructure complete with an eminent panel of multi-specialty physicians.”
One assumes this is the third party medical practitioners, given that the same web page states that only one clinic was performing the therapy in question. Yet, upon looking up the CCSVI Clinic for more information, I find this disclaimer:
“CCSVI Clinic is in the business of facilitating travel and hospitality only. Any and all medical services are provided by medical professionals who are not in the employ of CCSVI Clinic Inc…. CCSVI Clinic does not recommend, endorse or provide any specific test, analysis, products, or procedures of a medical nature.“
I’m sure more digging would bring forward additional “mischaracterizations, misinformation and misunderstanding” all of which originate from the now eerily quiet company itself.
Two minutes for obfuscation? It might take months or years to get clarity on this one.
Lisa Willemse
Latest posts by Lisa Willemse (see all)
- Toronto team tackling unmet need in cell manufacturing using synthetic biology - January 9, 2020
- Medicine by Design-funded researchers devise new strategy to improve the safety of cell therapies - February 22, 2019
- Right Turn: These three videos show why we should be impressed by our young stem cell researchers - November 18, 2016






Comments