In this month’s blog on addressing specific bioprocess and bioanalytical challenges to develop Cell and Gene Therapies (CGTs), we hear from Dr. Nick Timmins on quantifying performance of CGTs using cell yield, and some of the methods to measure it, as a metric of optimized manufacturing of CGTs. (SV)
Nick Timmins is VP, Process Science at BlueRock Therapeutics – a company focused on harnessing the power of induced pluripotent stem cells as a manufacturing platform for cell based medicines – leading the company’s manufacturing development efforts. Find more information about Nick here.
From quantifying the success or otherwise of a cell culture process, to evaluating promising new technologies or ensuring therapeutic product is available to those who need it at an acceptable cost, yield is an often overlooked, but incredibly important metric in cell and gene therapies. What does “yield” mean and what can it tell you? In the simplest form Yield = units out/units in. That is, for any one unit of a given input material, the number of units of the desired output is obtained.
For this discussion, let us assume that the output unit is a cell of a specific identity and function (X). The choice of input material is dependent on context, a common example for bio-based manufacturing being the quantity of substrate that is consumed (S). Thus, in our example one could express the yield as number of cells of type X obtained per gram of glucose consumed: ![]()
In a cell therapy context, it is often useful to express yield based on the number of input cells (e.g., number of cardiomyocytes obtained per input pluripotent stem cell from which they were differentiated). Unfortunately, it is common in the scientific literature (and presentations) that performance of such culture processes is reported only in terms of the percentage of desired cells present in the total population of cells obtained (e.g., %cTnT +ve cells as a measure of “how many” cardiomyocytes are obtained).
While important, such measures fail to account for the increase or decrease in cell numbers that occur. For example, a differentiation process (A) may provide a relatively pure population comprising 85 per cent of cell type X, but result in significant cell death where the total cell number is reduced by half, Y = (0.85 x 0.5) / 1 or 0.425. Conversely, a process (B) producing a 60 per cent pure population, but a five-fold net increase in total cell number, gives a yield of three.
If A and B consume equal quantities of resources (e.g., media, reagents, consumables, labour), process B may have a significant advantage due to the seven-fold higher yield relative to A.
This statement drives towards a key consideration for commercialization of new therapies and the ability to reach many individuals, i.e. manufacturing cost. The input units, or substrate, used to describe yield correspond to a cost that is incurred per unit output. This may be the cost of substrate, the cost incurred in generating input cells, or at the top level simply the number of cells obtained/per (total) $ input. This leads me to some questionable, but useful (and deliberately manipulative) math.
Let us say that![]()
That is, the “Product” one obtains is a function of what we put in, what we get out, the quality of what we get out, and how many $ were spent to obtain it. For a given therapeutic cell product, the quality that needs to be achieved is effectively fixed, and we can take it to be a constant. We can then write ![]() Cost is usually expressed in monetary terms, so
Cost is usually expressed in monetary terms, so ![]() From the information above, it is hopefully clear that yield
From the information above, it is hopefully clear that yield ![]()
is intrinsically linked to cost, as substrate, or input, and will have an associated monetary value. For example, the cost of cell culture media and supplements. We can then write ![]()
The cost term now dominates and is the major determinant of the magnitude of “Product.” Thus, it is important to minimize cost and an essential element to doing so is developing an understanding of what you get out, for what you put in. That little gem of circular logic takes us back to yield, and why it is an important metric to truly understand the performance of your method/process that could lead to the next game-changing therapy.
How can one “measure” yield? The change in viable cell quantity can be measured by direct cell counting using a variety of common laboratory methods (e.g., trypan blue exclusion or fluorescence-based cell counters), or using non-destructive technologies such as biomass measurement probes.
Measures of nutrient (e.g., glucose) or oxygen consumption, or metabolite generation (e.g., lactate) can also be used as surrogate measures of viable cell quantity. Where heterogeneous populations of cells are obtained, it is important to also quantify the fraction of the population corresponding to the target cell type. A common approach is to determine the fraction of cells expressing specific proteins that are characteristic of the desired phenotype (e.g., by flow cytometry). Measures of cell morphology, optical properties, or gene expression might also be used. Regardless of the analytical method, it is critical that a quantitative definition of the target cell type is established for such measurements to be meaningful.
Measuring the change in “input” is dependent on the basis used. Where the input corresponds to a starting cell type, the approaches described above apply. If bulk medium consumption is the relevant parameter of interest, this can be determined from measurements of volume or mass. Where specific media components are used as a basis, a variety of analytical technologies are relevant depending on the nature of that specific component.
It is important that choice of “input” and corresponding analytical technology is made with careful consideration of what it is that you want to learn from yield determination, and what decisions are to be informed by these learnings.
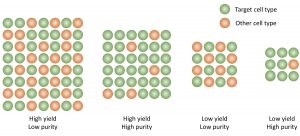
While it is common to see percentage purity as a measure of culture success, this fails to describe how productive a culture has been in terms of target cell yield. Consider the situations above where the same number of input cells was used to achieve each outcome. While the two scenarios on the left differ greatly in percentage purity, the yield of target cells per input cell is the same. While the two right-hand scenarios produce much fewer cells in total, and differ in purity, the yield of target cells is equal. (Credit Nick Timmins, BlueRock Therapeutics and Carmen Wong, CCRM)
Sowmya Viswanathan
Latest posts by Sowmya Viswanathan (see all)
- Raman spectroscopy for monitoring therapeutic cell manufacturing – Part 2 - March 28, 2019
- Without new funding, key part of Canada’s stem cell research ecosystem to close - February 28, 2019
- Raman spectroscopy for monitoring therapeutic cell manufacturing – Part 1 - September 6, 2018






Comments