In many regenerative medicine strategies, we know that one strategy is usually not enough. Stimulating regeneration in any tissue is a complex, multifaceted problem involving the coordination of many biological signals. Yet what if we could deliver a therapeutic, like a drug or cells, to the body in such a precisely controlled manner that we knew exactly when it would be released, and for how long? Or if we could deliver multiple therapeutics at once, with their release into the tissue choreographed like well practiced dancers?
Researchers are hard at work to develop new ways to control the delivery of therapeutics using biomaterials. Biomaterials can be used to localize therapeutics to the site of action in a way that cannot be achieved by delivery to the bloodstream (systemic delivery). They can protect drugs from degradation by the body, reduce the immune response, and provide cells with a scaffold on which to grow and survive. Also, by enabling delivery right to the site of action, biomaterials diminish potentially harmful side-effects of systemic delivery. (For more information on research into biomaterials, please see my other Signals posts here and here.) Despite the many benefits of biomaterials, one of the problems that researchers are still grappling with is that once the biomaterial is implanted into the body, we can no longer control what happens to its payload.
To understand why this is important for regenerative medicine, consider the example of heart regeneration following infarction (tissue death due to inadequate blood supply from an obstruction). We may want to deliver a biomaterial containing both a drug and cells, but not necessarily introduced into the tissue at the same time: the drug may be released quickly to make the environment of the injured heart less hostile to the cells, which would then be released from the material after the drug has taken effect.
New work, published in Nature Chemistry, has demonstrated a method to control the release of multiple therapeutics by adapting the language of computers, with very interesting possibilities for regenerative medicine. The researchers ingeniously combine biology, chemistry and computational engineering to create smart biomaterials for drug or cell delivery. They describe their system as capable of “biocomputation,” which “represents the ability to simultaneously sense multiple biologically presented inputs and follow a user-programmed Boolean logic-based algorithm to provide a functional output.”
Boolean logic is a basic theory that underlies much of our modern technology. In short, it operates on the assumption that a statement is either true or false. If statement A is true (“YES”) then it is assigned 1, and if it is false it is assigned 0. The next level involves two statements, A and B. An “OR” means that of A and B one must be true, and an “AND” means that A and B must both be true. These true/false statements act as gates; if a statement is true then the desired function is performed.
To translate the language of computers to chemistry, the authors converted the 1s and 0s into chemical structures. Each uniquely tailored region of the biomaterial, called a crosslinker, will degrade in response to different cues in the surrounding environment, releasing the payload of drugs or cells. The researchers used three different crosslinkers, similar to Boolean logic: “YES” which contains one site of degradation that will respond to an environmental cue and cause the material to degrade, “OR” which contains two different sites of degradation that respond to two distinct environmental cues, so only one needs to be present for the whole material to degrade, and “AND” which also contains two different chemical sites of degradation that are connected in such a way that both have to degrade to make the whole material degrade, i.e., both environmental cues must be present to get degradation and therapeutic release. Those are the simplest ways to make the biomaterial, but it can get even more complex, with multiple combinations of “YES” “OR” and “AND” in the chemical structure of the material to create precise, higher order responses.
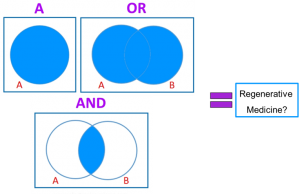
Figure Caption: New research from Nature Chemistry demonstrates that Boolean logic can be used to tailor drug and cell delivery for future regenerative medicine applications.
Not limited to just theorizing about the use of Boolean logic for biomaterials, the researchers also demonstrated its efficacy using a simple cell culture assay. First, they delivered a cancer drug bound in a biomaterial that required two environmental inputs to release the drug (an “AND” chemical crosslinker). They showed that significantly more cancer cells were killed with their system as opposed to simply adding the drug to the cells or encapsulating the drug in a biomaterial without the responsive crosslinkers. The researchers also showed that they could encapsulate live cells in a biomaterial that uses changes in UV light wavelength to degrade the material and release the cells. When they experimentally exposed cells to different wavelengths, different populations of cells could be released in response.
So far, this technology has been demonstrated in a dish with synthetically generated cues such as changes in light or chemical concentration and has yet to be tested in an animal model with natural environmental cues. The researchers, however, theorize that the cues found in the body should be able to produce similar results if the chemistry is tailored accordingly. This work is a unique combination of an old mathematical algorithm and chemical structures to conduct complex and smart release of multiple therapeutics.
Samantha Payne
Latest posts by Samantha Payne (see all)
- Growing pains for the regenerative medicine industry - August 29, 2018
- It’s electrifying! Cell reprogramming using electromagnetic fields - May 24, 2018
- Adapting the language of computers for regenerative medicine - February 16, 2018






Comments