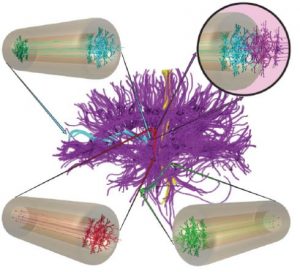
A schematic of the micro-TENNS technology. A long hollow tube composed of a biomaterial is filled with neurons at one end, which extend long processes through the tube. The construct is transplanted into the brain where it can interface with host neurons to repair long-distance pathways. Image modified from Struzyna et al., Neural Regeneration Research 2015.
The brain is one of the most complex and delicate organs of the body, with very little capacity to regenerate itself. As such, any disease or injury it sustains is a challenge for regenerative medicine researchers to design effective strategies. Therapeutics need to be minimally invasive so the brain is not damaged further, and they need to avoid causing a foreign body response by the immune system. The organization of the brain tissue is also extremely complex, with discreet functional and anatomical areas composed of a myriad of cell types that are difficult to duplicate with regenerative strategies.
Luckily, many researchers around the world are looking at ways that biomaterials can help to regenerate and treat brain disease and injury. The term biomaterial encompasses a host of materials that researchers often choose to work with because they are customizable and compatible with the tissues of the body. The focus of this blog is the work done by the Cullen Lab, at the University of Pennsylvania, where researchers have designed something they call micro-tissue engineered neural networks (micro-TENNs) that combine biomaterials and cells in an unusual way for delivery into the brain. These constructs are essentially tiny tubes of organized neurons inside a biomaterial shell, around the diameter of a human hair and up to several centimeters in length.
To construct these devices, the Cullen Lab first makes an outer tube of biocompatible hydrogel that can be filled with components of the extracellular matrix (ECM) and different types of neurons that are found in the brain: excitatory or inhibitory neurons, or dopaminergic neurons important for Parkinson’s. They then culture the neurons in the tubes, which will guide the axons of the neurons along a specific path, creating long distance networks that are similar to how they would be organized in the brain. When the cells have created these networks, the tubes are then ready to be implanted into the brain. Micro-TENNS are so small they can be injected through a syringe into the tissue, which helps to minimize damage to the brain.
There are two main applications for micro-TENNS that the Cullen Lab is pursuing. The first is repair of the brain. Some areas of the brain that get damaged due to stroke, traumatic brain injury, or neurodegenerative diseases contain long-distance tracts for neurons that are difficult to repair with conventional transplantation strategies. The hope is that because the micro-TENNS is customizable, any type of neuron can be used to create these organized tubular structures, which can then be used in a variety of applications. In a 2015 publication the Cullen Lab showed that a micro-TENNS construct containing cortical neurons can be injected through a syringe as a whole structure into rat brains, lasting for 28 days and physically bridging cells to establish connections in the brain. They improved upon the original design with a publication last year by demonstrating that adding a layer of a stiffer material to the outside of the tube allows the insertion of the micro-TENNS directly into the brain, without the help of a needle, further minimizing any disruption to the brain tissue.
The next step for the Cullen Lab, outlined in a review paper from earlier this year, is to move away from replacing cells in the brain and towards measuring and manipulating brain function. Current methods to measure the input and output of the brain require either invasive procedures like microelectrode implants, or non-invasive techniques like light that cannot penetrate to the deeper structures of the brain.
The Cullen Lab hopes to apply their micro-TENNS technology toward making “living electrodes.” The idea is that the micro-TENNS filled with neurons can be inserted into the brain, but with a portion open to the surface. The neurons inside can form connections with the host cells at the deep end of the construct, while various stimulants can be applied at the surface of the construct that will propagate down the tube to affect the host cells. The Cullen Lab envisions that this technology can be used to target very localized populations of cells in the brain and suggest a variety of interesting applications depending on where in the brain it is inserted, such as inhibiting seizure circuitry, stimulating cells to release neurotransmitters for Parkinson’s, and aiding in learning and memory. Although these applications have not yet been tested in animal models, it will be exciting to see what the next five years brings for the micro-TENNS technology.
Samantha Payne
Latest posts by Samantha Payne (see all)
- Growing pains for the regenerative medicine industry - August 29, 2018
- It’s electrifying! Cell reprogramming using electromagnetic fields - May 24, 2018
- Adapting the language of computers for regenerative medicine - February 16, 2018






Comments